|
Putting these pieces together gives the name carbon tetrachloride for this compound. At ordinary ambient temperatures, it is a green-brown ionic solid consisting of iron (II) cations Fe2+. 3, that occurs naturally as the mineral siderite. The second element, chlor ine, becomes chlor ide, and we attach the correct numerical prefix (“tetra-”) to indicate that the molecule contains four chlorine atoms. Iron (II) carbonate, or ferrous carbonate, is a chemical compound with formula FeCO. The name begins with the name of the first element-carbon. In their short piece of article, know more about the carbonate. The main group of elements used as carbonates are alkali and alkaline earth metals. Carbonate is a salt of carbonic acid possessing the molecular formula CO 32. Let us practice by naming the compound whose molecular formula is CCl 4. formula M2(CAACCO2)n (58) with reduced CO2 units which are structurally analogous to the carbonite anion CO22. The fizz in the club soda, the foaming effect in the washing soda are all due to carbonate ions. Inorganic carbonates comprise many minerals ( see carbonate mineral) and are the. 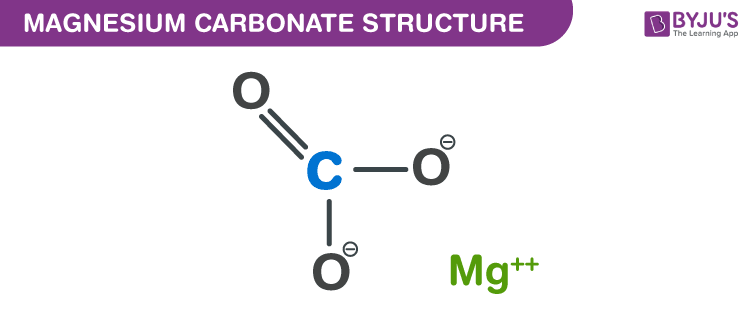
It is found naturally in rocks, soil, water, plants, and animals. Cobalt is a metallic element with the atomic number 27. It occurs naturally as the mineral spherocobaltite and is used in pottery glazes. The inorganic carbonates are salts of carbonic acid (H 2 CO 3 ), containing the carbonate ion, CO 2/3-, and ions of metals such as sodium or calcium. Cobalt (II) carbonate is a chemical compound of cobalt. What is the correct formula for the acid that. 
*This prefix is not used for the first element’s name. carbonate, any member of two classes of chemical compounds derived from carbonic acid or carbon dioxide ( q.v. It is an ion, not an element, nor should it be mistaken for the covalently bonded compound CO2, carbon dioxide. \): Numerical Prefixes for Naming Binary Covalent Compounds Number of Atoms in Compound In this video well write the correct formula for Manganese (IV) carbonate : Mn(CO3)2.To write the formula for Manganese (IV) carbonate we’ll use the Periodi.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
